ADVERTISEMENT
- Rozovsky wins prestigious NSF Early Career Award
- UD students meet alumni, experience 'closing bell' at NYSE
- Newark Police seek assistance in identifying suspects in robbery
- Rivlin says bipartisan budget action, stronger budget rules key to reversing debt
- Stink bugs shouldn't pose problem until late summer
- Gao to honor Placido Domingo in Washington performance
- Adopt-A-Highway project keeps Lewes road clean
- WVUD's Radiothon fundraiser runs April 1-10
- W.D. Snodgrass Symposium to honor Pulitzer winner
- New guide helps cancer patients manage symptoms
- UD in the News, March 25, 2011
- For the Record, March 25, 2011
- Public opinion expert discusses world views of U.S. in Global Agenda series
- Congressional delegation, dean laud Center for Community Research and Service program
- Center for Political Communication sets symposium on politics, entertainment
- Students work to raise funds, awareness of domestic violence
- Equestrian team wins regional championship in Western riding
- Markell, Harker stress importance of agriculture to Delaware's economy
- Carol A. Ammon MBA Case Competition winners announced
- Prof presents blood-clotting studies at Gordon Research Conference
- Sexual Assault Awareness Month events, programs announced
- Stay connected with Sea Grant, CEOE e-newsletter
- A message to UD regarding the tragedy in Japan
- More News >>
- March 31-May 14: REP stages Neil Simon's 'The Good Doctor'
- April 2: Newark plans annual 'wine and dine'
- April 5: Expert perspective on U.S. health care
- April 5: Comedian Ace Guillen to visit Scrounge
- April 6, May 4: School of Nursing sponsors research lecture series
- April 6-May 4: Confucius Institute presents Chinese Film Series on Wednesdays
- April 6: IPCC's Pachauri to discuss sustainable development in DENIN Dialogue Series
- April 7: 'WVUDstock' radiothon concert announced
- April 8: English Language Institute presents 'Arts in Translation'
- April 9: Green and Healthy Living Expo planned at The Bob
- April 9: Center for Political Communication to host Onion editor
- April 10: Alumni Easter Egg-stravaganza planned
- April 11: CDS session to focus on visual assistive technologies
- April 12: T.J. Stiles to speak at UDLA annual dinner
- April 15, 16: Annual UD push lawnmower tune-up scheduled
- April 15, 16: Master Players series presents iMusic 4, China Magpie
- April 15, 16: Delaware Symphony, UD chorus to perform Mahler work
- April 18: Former NFL Coach Bill Cowher featured in UD Speaks
- April 21-24: Sesame Street Live brings Elmo and friends to The Bob
- April 30: Save the date for Ag Day 2011 at UD
- April 30: Symposium to consider 'Frontiers at the Chemistry-Biology Interface'
- April 30-May 1: Relay for Life set at Delaware Field House
- May 4: Delaware Membrane Protein Symposium announced
- May 5: Northwestern University's Leon Keer to deliver Kerr lecture
- May 7: Women's volleyball team to host second annual Spring Fling
- Through May 3: SPPA announces speakers for 10th annual lecture series
- Through May 4: Global Agenda sees U.S. through others' eyes; World Bank president to speak
- Through May 4: 'Research on Race, Ethnicity, Culture' topic of series
- Through May 9: Black American Studies announces lecture series
- Through May 11: 'Challenges in Jewish Culture' lecture series announced
- Through May 11: Area Studies research featured in speaker series
- Through June 5: 'Andy Warhol: Behind the Camera' on view in Old College Gallery
- Through July 15: 'Bodyscapes' on view at Mechanical Hall Gallery
- More What's Happening >>
- UD calendar >>
- Middle States evaluation team on campus April 5
- Phipps named HR Liaison of the Quarter
- Senior wins iPad for participating in assessment study
- April 19: Procurement Services schedules information sessions
- UD Bookstore announces spring break hours
- HealthyU Wellness Program encourages employees to 'Step into Spring'
- April 8-29: Faculty roundtable series considers student engagement
- GRE is changing; learn more at April 15 info session
- April 30: UD Evening with Blue Rocks set for employees
- Morris Library to be open 24/7 during final exams
- More Campus FYI >>
11:02 a.m., Sept. 15, 2010----The Clinical Laboratory Improvement Amendments (CLIA), passed by the federal government in 1988, regulate collection and analysis of fluid and tissue samples in the diagnosis and treatment of illness.
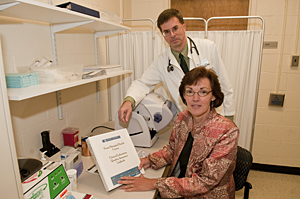
“On the University of Delaware campus, CLIA governs every Wellness Center screening for cholesterol or glucose, every lab test done in the new Nurse Managed Health Center, and every diagnostic lab test -- from strep throat to mononucleosis -- done in the Student Health Center,” says Mary Ann McLane, professor in UD's Department of Medical Technology.
According to McLane, both the Student Health Center and the Department of Medical Technology hold CLIA federal certificates authorizing them to support and provide training for any diagnostic lab testing done at the University. The Nurse Managed Health Center was recently added to the Medical Technology CLIA certificate, so that NMHC personnel can perform urinalysis and blood glucose testing in McDowell Hall and cholesterol screenings at the Multiple Sclerosis Clinic in McKinly Lab.
“Significant sanctions are possible if such diagnostic testing is done without a CLIA certificate in place,” she says. “Staff members in these clinics ensure that personnel and quality assurance requirements are met whenever clinical lab testing is done on campus.”
Medical technology faculty are involved with a wide range of clinical laboratory testing activities on campus, including providing cholesterol and glucose testing during Alumni Weekend and offering annual seminars to graduate students who conduct Wellness Center cholesterol and glucose screenings.
These faculty members will also be pivotal to the addition of diagnostic laboratory testing at the health sciences complex on UD's new science and technology campus within the next few years.
In addition, as part of UD's participation in the Eastern Pennsylvania-Delaware Geriatric Education Center, med tech faculty are developing training videos for primary care staff to improve understanding of the aspects of diagnostic testing that are unique to older adults.
McLane emphasizes that the CLIA regulations do not apply to samples collected for research purposes. However, she says, department faculty are willing to assist anyone at UD who is planning to do such testing.
The University community is encouraged to seek the expertise of the Department of Medical Technology (831-2849) in the planning stages for any project involving diagnostic laboratory testing of human subjects.
A brief overview of the role of the medical laboratory scientist in healthcare is available at this YouTube site.
Article by Diane Kukich
Photo by Doug Baker


