CHEM-342 INTRODUCTION TO BIOCHEMISTRY
MIDTERM EXAMINATION - PART I (Individual work)
Thursday 23 March 1995, 7 - 10 p.m.
H. B. White - Instructor
Read this before you turn the page.
1. You should have plenty of time to complete this examination. Therefore, take your time, organize your thoughts, and write "not that you may be understood but rather that you can not possibly be misunderstood." Stream of consciousness answers are rarely well organized. I will be looking for clear, well thought out answers that properly use examples from the course and personal experience.
2. PART I (75 points) You will have up to 90 minutes to answer Part I which contains four questions. You may use your course notes, handouts, and reader. However, texts or other reference books cannot be used on this section. If you finish Part I early, you may leave the room and relax until Part II begins around 8:30 p.m.
3. PART II (25 points) Problem 4 from Part I will be answered again in Part II but with the benefit of group discussion and with access to reference books. Each group will hand in a single answer to be graded and the group grade will be added to your score on Part I. If you do not agree with your group's answer, you may hand in a separate answer which will be graded and used in place of the group grade.
GOOD LUCK
1. (20 points) Section 11 of Stokes' article describes an experiment that we deciphered and demonstrated in class as listed in the procedure below. Stokes (and we) observed the behavior of the colored solute at each step.
|
PROCEDURE |
OBSERVATIONS |
| a. Mix a dilute solution of blood with an equal volume of diethyl ether. | A two phase system formed with a clear organic phase above and a pink aqueous solution below. |
| b. Add glacial acetic acid and mix gently. | The aqueous solution turned brown and the brown color transferred to the upper organic layer leaving a white precipitate in the aqueous layer. |
| c. Wash the ether phase several times with water. | After several washes the brown color formed a dark layer near the water-ether interface. |
| d. Add a few drops of ammonium hydroxide. | The ether layer became clear and the color transferred to the aqueous layer. |
For each step in the above procedure:
You may continue your answer onto the back of this page.
2. ( 25 points) Assume you were writing a textbook in 1925 and wanted to give an up-to-date (through Conant's article) and accurate summary of what was then known about hemoglobin. Write a concise paragraph or two that a typical undergraduate science major would understand. Although textbooks then were not as profusely illustrated as they are now, consider what illustrations, equations, or chemical formulae you would use to conceptualize the ideas in your text.
3. (25 points) Although the reactions of hemoglobin that Conant studied were outside the body, his work on the reversible formation of methemoglobin from hemoglobin has physiological relevance. Some reactions in the body produce hydrogen peroxide, which can react with hemoglobin to form methemoglobin. Methemoglobin cannot bind and transport oxygen; consequently, processes have evolved to decrease the formation of methemoglobin and also to convert it back into functional hemoglobin that can bind oxygen. For example, red blood cells contain an abundance of catalase, an enzyme that destroys hydrogen peroxide by converting it to water and oxygen. (This enzyme causes the bubbles of oxygen to form when hydrogen peroxide is put on a wound.) Despite the protective presence of catalase, methemoglobin forms naturally and constitutes about 0.5% of the total hemoglobin in your body. Another enzyme in red blood cells converts methemoglobin back to hemoglobin. Humans genetically deficient in this enzyme can have 30% methemoglobin in their red blood cells. Treatment of these patients with reducing agents like ascorbic acid (Vitamin C) or methylene blue considerably lowers the amount of methemoglobin in their blood.
Using chemical formulae, abbreviations, and equations, construct a diagram that includes the interconnection of the various underlined molecules in the reactions described above.
Bonus (5 points) Several decades ago the first patient lacking catalase was discovered by accident by a Japanese surgeon. How was the discovery made?
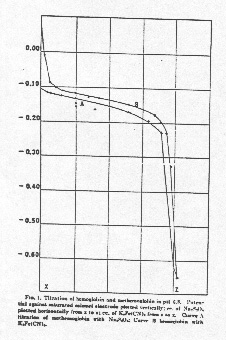
4. (10 points) Figure 1 below from Conant's article shows the changes in electrochemical potential during anaerobic, reversible titrimetric reduction of methemoglobin. What would this figure look like if a seal broke permitting air toflood the system one third of the way into the initial titration labeled "A" and the experiment were continued to completion? Draw your predictions on the figure and be as precise as you can. Explain your answer qualitatively and quantitatively. Would the results be any different if carbon monoxide, rather than air, were introduced?

When you are done with Part I, you may hand it in and relax in the hall until Part II begins around 8:30 p.m.
CHEM-342 INTRODUCTION TO BIOCHEMISTRY
MIDTERM EXAMINATION - PART II (Group work)
Thursday 23 March 1995
H. B. White - Instructor
In Part II, your group will reanswer Question 4 from Part I. You may use any of the reference books available. Your group should come to consensus on a single group answer which constitutes 25% of this midterm examination. However, if after full discussion, your answer does not agree with that of the rest of your group, you may submit you answer for separate grading and separate credit.
The examination should end around 9:30 p.m. Any group or individual that needs more time can stay longer, within reason. Class will not meet tomorrow morning.
4. (25 points) Figure 1 below from Conant's article shows the changes in electrochemical potential during anaerobic, reversible titrimetric reduction of methemoglobin. What would this figure look like if a seal broke permitting air to flood the system one third of the way into the initial titration labeled "A" and the experiment were continued to completion? Draw your predictions on the figure and be as precise as you can. Explain your answer qualitatively and quantitatively. Would the results be any different if carbon monoxide, rather than air, were introduced?
