CHEM-342
Introduction to Biochemistry
Group Members________________________
Mid-term Examination - Group
Part
Wednesday, 21 March 2001
H. B. White - Instructor
Important - Please read this before you turn the page.
-
Write your names or group number on each page.
-
You may refer to your notes, course reader, handouts, or
graded homework assignments. Reference books in the course library may
be consulted briefly and returned..
-
Please read the question carefully and make sure that you
have thought it through with everyones input before converging
on a solution.
1. Like Zinoffsky, both Conant and Svedberg
& Fåhraeus studied equine hemoglobin.
A. (2 points) Figure 1 of
Conants article shows the reversible electrochemical titration of hemoglobin.
What are the colors of his hemoglobin solutions at the end points of the
titrations with ferricyanide and dithionite (hydrosulfite), respectively?
B. (2 points) Svedberg & Fåhraeus
determined the molecular weight of two different forms of hemoglobin. What
were the colors of each of these forms?
C. (3 points) Would the color of the
hemoglobin solution be of any importance to the experiments by either Conant
or Svedberg & Fåhraeus?
2. (3 points each) State clearly in
one sentence, the major conclusion/significance for each article.
A. Conant:
B. Svedberg & Fåhraeus:
3. (6 points) Dr. Essigsaure
is trying to repeat Conants experiment but he hasnt fully mastered the
technique. Halfway through a beautiful electrochemical titration with dithionite,
his system developes a leak in which oxygen enters the system before he
reseals it. Never the one to waste an opportunity, he continues the titration
to the end point with the results shown below: Estimate the amount of oxygen
(relative to total hemoglobin) that leaked into the cell.

Volume of standardized dithionite
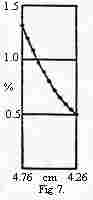
4. (6 points) Dr. Essigsaure
loves to play in the lab but avoids calculations. A friend of his studies
a type of worm that is "blood red," thinks they contain hemoglobin, and
wants to find out its molecular weight. Every ready Essigsaure, cranks
up the old analytical ultracentrifuge following Svedbergs instructions
and soon discovers that the protein is much larger than he expected. He
keeps lowering the speed of the rotation until the concentration curve
exactly matches the one shown in Svedberg and Fåhraeus article.
After many trials in which a less persistent but more mathematically inclined
scientist would have simply calculated the molecular weight or the appropriate
rotational velocity, Dr. Essigsaure found that when the rotational speed
was about 25% that of Svedberg and Fåhraeus, his concentration profile
was as shown. What is the approximate molecular weight of the worm hemoglobin?
What assumptions do you have to make?
Bonus Question (1 point) What does "Essigsaure" mean in German?
Return to Hal
White's Home Page or Departmental
Home Page.
Last updated: 21 March
2001 by Hal White
Copyright 2001, Department
of Chemistry and Biochemistry, University of Delaware