
Arnold L. Rheingold
Professor of Chemistry
 |
Arnold L. Rheingold Professor of Chemistry |
| Summary | Research | Education | Selected Publications | Membership in Professional Societies |
| Department of Chemistry and Biochemistry
University of Delaware Newark, DE 19716 (302) 831-8720 (office) (302) 831-6335 (FAX) e-mail: arnrhein@udel.edu |
 |
Inorganic Chemistry. Main-group and transition-metal organometallic chemistry. Gas-phase cluster synthesis. Thermoelectic materials. Single-crystal structural determinations.
Clusters are large molecules usually built around tightly constructed frameworks. Because they are complex yet molecular they can serve as precise precursors to a myriad of solid-state compositions including ceramics, semi- and superconductors, photoresp onsive agents and thermoelectric materials. Cluster structures formed from a combination of main-group and transition-metal elements are especially attractive subjects for study due to their novelty and potential application in thermoelectric materials (TEM ). TEM's either change temperature on passage of an electrical current or generate electricity when exposed to a thermal gradient. Examples of potentially important applications include non-mechanical domestic refrigeration and converting an automobile' s exhaust heat into electricity eliminating the alternator. A thin film of a TEM could be used to create a self-cooling computer chip.
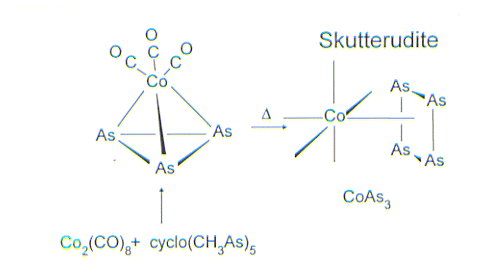
One promising area for the fabrication of TEM's uses elements from the late transition metals from groups 8, 9 and 10 in combination with the heavier main-group elements from groups 14, 15 and 16. An example is cobalt arsenide, CoAs3, which occurs naturally as the mineral skutterudite. Using the strategy of isolobal replacements for individual atoms or groups of atoms in skutterudite, we are exploring the synthesis and electrical property characterization of new compositions of matter with enhanced thermoelectric efficiencies.
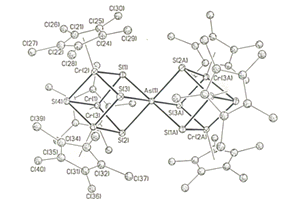
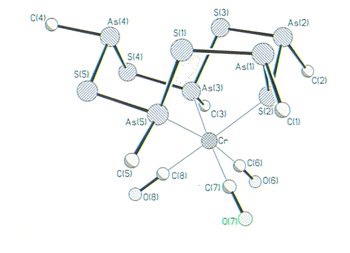
Clusters such as [Co(CO)3]As3 and FeNi(CO)6As2 thermally decompose to form novel TEM's. Other interesting examples of new cluster designs include the Cr/As cluster shown below that has the highly unusual proper ty of being a molecular with ferromagnetically coupled spins.
With an intense interest in solid-state materials, we have also become proficient in use of X-ray Crystallography for structural characterization and have established collaborations with chemists from about thirty universities and corporations in the study of new crystalline materials. Many of these collaborations are with leaders in their fields which affords an extraordinary opportunity to train crystallographers using examples from some of the most exciting research areas including nanostructured materials, molecular self assembly, bioinorganic chemistry and asymmetric synthesis.
 |
Case Western Reserve University. A.B., 1962 in chemistry; M.S. 1963 in inorganic chemistry. University of Maryland. Ph.D., 1969 in inorganic chemistry. Virginia Polytechnic Institute. Postdoctoral Study. Professor Raymond Dessy. 1969-70. SUNY-Buffalo. Sabbatical Leave. Professor Melvyn Churchill. 1980-81. |
| University of Delaware Home Page |  | |
| Chemistry Department | ||
| X-ray Crystallography Home Page | ||
| Back to Top |